
Use Cases
Track FDA regulations
Features
Categories
Overview
Compliance officers and legal teams in healthcare organizations have to stay current on changing regulations, FDA approvals, and industry standards across multiple jurisdictions. Perplexity can help by aggregating regulatory updates, compliance requirements, and recent enforcement actions. It can then create compliance dashboards and regulatory horizon reports.
Your search
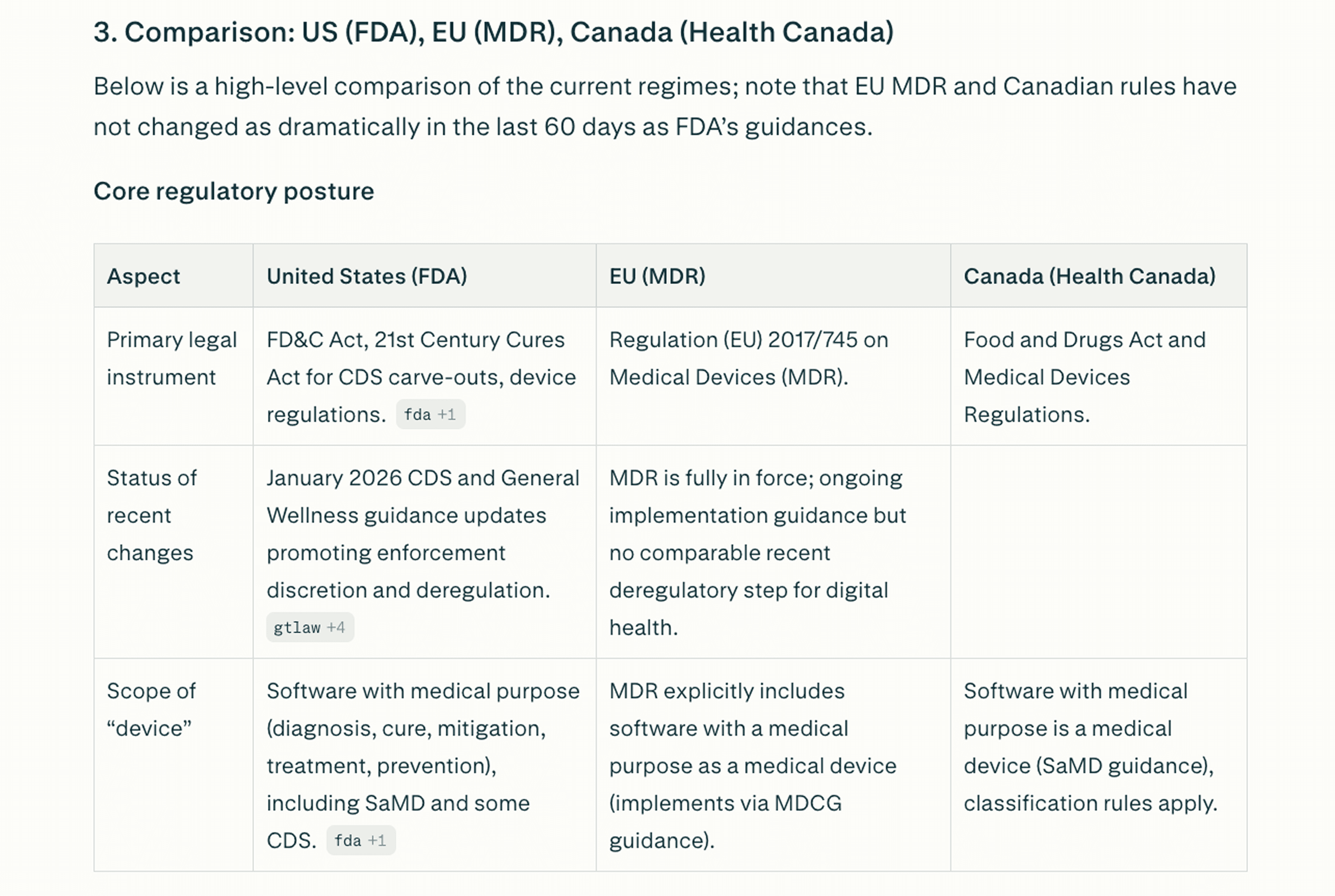
What are the latest FDA regulations affecting digital health applications launched in the last 60 days? Include any new guidance documents, enforcement actions, and pending rule changes. Compare requirements across US, EU (MDR), and Canada. Flag any that affect telemedicine platforms specifically.
Perplexity's answer
Perplexity surfaces:
Recent FDA guidance documents with effective dates and key requirements
Enforcement actions taken against companies (with violation details)
Pending rules and comment periods still open
Regional comparison showing differing requirements for US, EU, and Canada
Specific impacts on telemedicine workflows and data handling
The report can be exported as a detailed PDF for your legal team, saved as a structured database of requirements for your compliance checklist, or shared as an interactive dashboard that auto-updates monthly. You can refine by asking follow-ups like "Which of these apply to our specific product category?" or "What's our implementation timeline for EU MDR compliance?"
Tips
Specify your product category: "mobile health apps," "wearables," "clinical decision support systems"
Request timeline clarity: "What's the deadline for compliance?" and "Are there grace periods?"
Ask comparative questions: "Which jurisdiction has the strictest requirements for data storage?"
Track enforcement patterns: "What are the most common violations in our space?"
Request proactive monitoring: "Alert me to any new guidance affecting [product type]"
